Lewis Structure Drawing Practice
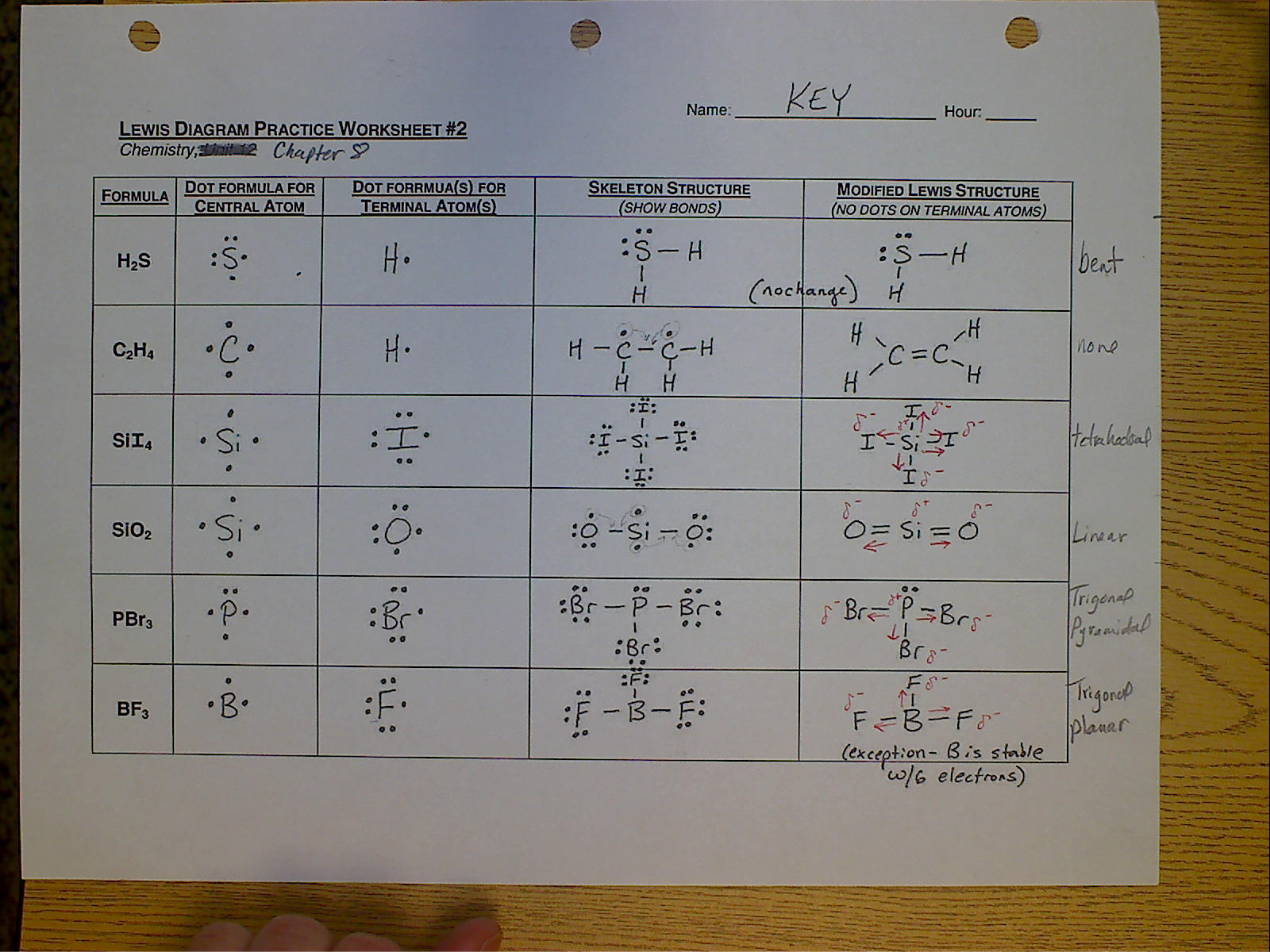
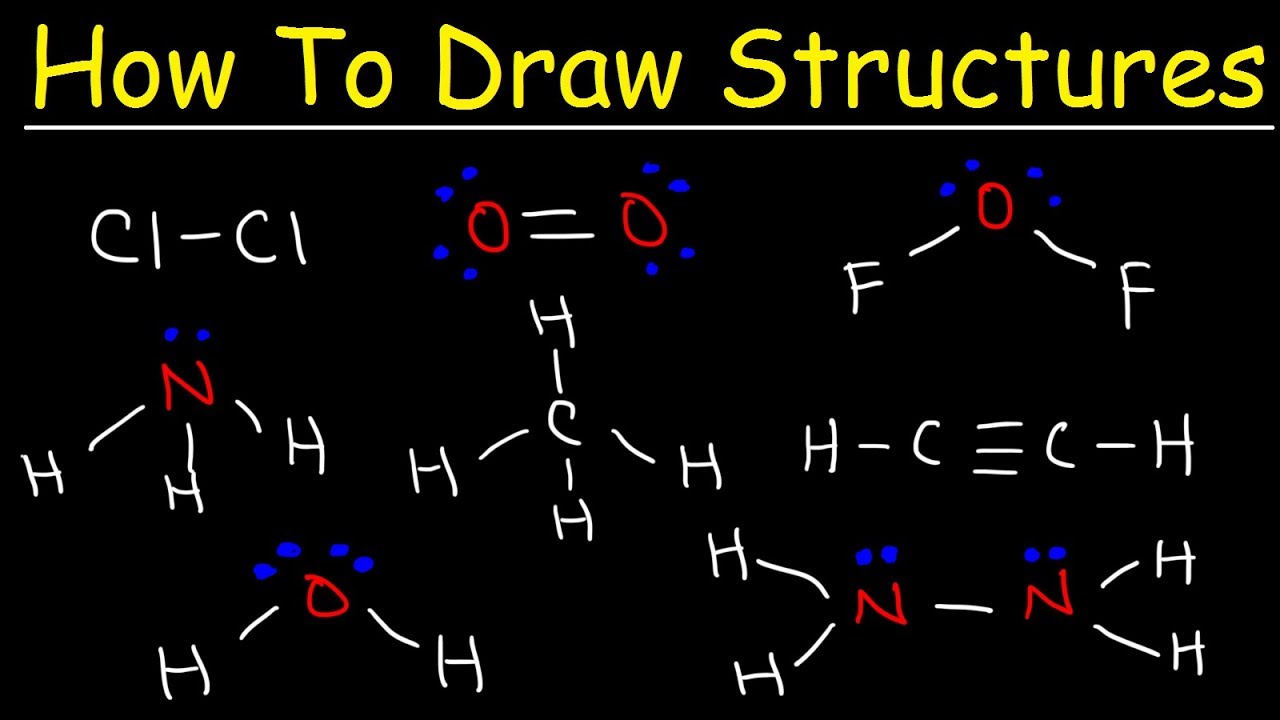
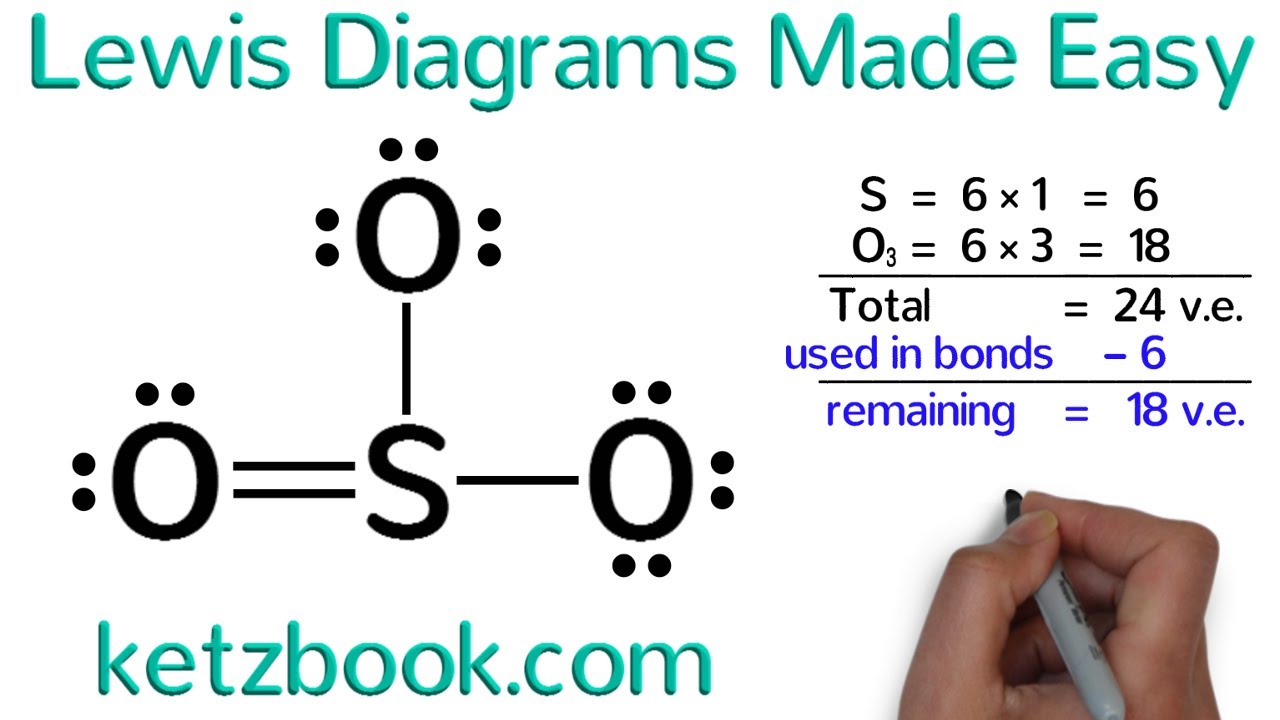
Lewis Structure Drawing Practice - One line is a single bond with 2 bonding electrons, two lines is a double bond with 4 bonding electrons, and three lines. Reference the “how to draw a lewis dot structure” for a step by step guide. Web learn for free about math, art, computer programming, economics, physics, chemistry, biology, medicine, finance, history, and more. Web drawing lewis structures for molecules with one central atom: Which of the following correctly completes the lewis diagram of ethanethiol? Complete the lewis structures of these molecules by adding multiple bonds and lone pairs. Ch 2 br 2 d. Web practise drawing the lewis structure of molecules using the exercises below. This video will explain how to find the formal charges. Web this online quiz is intended to give you extra practice in identifying and drawing lewis dot structures as well as predicting ion formation. Web write the lewis structures for carbon tetrachloride and phosgene. (there are molecules, like o 2, which have unpaired electrons even though they could all be paired, but you can't predict that with lewis structures, so assume they are all paired.) Web this online quiz is intended to give you extra practice in identifying and drawing lewis dot structures as well as predicting ion formation. Web the electrons in the outermost shell are called the _____ electrons. Web here are the steps to draw a lewis structure. Web drawing lewis structures for molecules with one central atom: See the following lewis dot structure diagrams for a few covalent compounds. You can usually return to this assignment anytime during the course, even after its due date. Web learn for free about math, art, computer programming, economics, physics, chemistry, biology, medicine, finance, history, and more. Write the correct skeletal structure for the molecule. Web practice drawing lewis structures with answers and explanation. Then name their electron arrangement, shape, and bond angles. In short, remember these steps for determining the lewis structure: Web examples for drawing lewis structures for covalent bonds. Draw a lewis structure for each of the following:d. See the following lewis dot structure diagrams for a few covalent compounds. Draw the lewis structure, then id shape and polarity. Answer the following questions and check your answers below. Be sure you know how to draw correct lewis dot structures and are able to correctly predict the electronic arrangement and molecular geometry before going on to the lab assignment.. Web the skeletal structure of ethanethiol is shown below. Draw the lewis structure, then id shape and polarity. The lewis structure rules and a periodic table while doing this exercise. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web draw a lewis structure for each of the. Web practice drawing lewis structures with answers and explanation. Web lewis structures practice worksheet draw the lewis structures for each of the following molecules. Web draw a lewis structure to answer a question. Then name their electron arrangement, shape, and bond angles. Web drawing lewis structures for molecules with one central atom: Answer the following questions and check your answers below. Add/replace, erase, or move atoms. The example is for the nitrate ion. Reference the “how to draw a lewis dot structure” for a step by step guide. In short, remember these steps for determining the lewis structure: Web this online quiz is intended to give you extra practice in identifying and drawing lewis dot structures as well as predicting ion formation. Draw the lewis structure for: Draw a lewis structure for each of the following:d. Draw the lewis dot structures for each of the following molecules: Write the correct skeletal structure for the molecule. Add/replace, erase, or move atoms. Web here are the steps to draw a lewis structure. All electrons should be in lone pairs or bonding pairs. Web drawing lewis structures for molecules with one central atom: Do not add any more atoms. Draw the lewis dot structure for each of the following polyatomic ions: All electrons should be in lone pairs or bonding pairs. Lewis diagram of the cyanide ion (cn⁻) Determine if the molecule is polar or nonpolar. Web examples for drawing lewis structures for covalent bonds. Lewis diagram of the cyanide ion (cn⁻) Web practice drawing lewis structures. Reference the “how to draw a lewis dot structure” for a step by step guide. Draw the lewis structure, then id shape and polarity. See the following lewis dot structure diagrams for a few covalent compounds. A periodic table will be available for the exam, but the list of rules will not be available, so this is a chance to practice using the rules to help you. This quiz aligns with the following ngss standard(s): The lewis structure rules and a periodic table while doing this exercise. Which of the following correctly completes the lewis diagram. One line is a single bond with 2 bonding electrons, two lines is a double bond with 4 bonding electrons, and three lines. See the following lewis dot structure diagrams for a few covalent compounds. Draw the lewis dot structures for each of the following molecules: Web practise drawing the lewis structure of molecules using the exercises below. Web this online quiz is intended to give you extra practice in identifying and drawing lewis dot structures as well as predicting ion formation. For the following molecules or ions (where the central atom is underlined): Web draw a lewis structure to answer a question. Web here are the steps to draw a lewis structure. The lewis structure rules and a periodic table while doing this exercise. Web when you draw the lewis structure, make all the electrons paired unless there is an odd number of electrons. Draw a lewis structure for each of the following:c. All electrons should be in lone pairs or bonding pairs. Web draw a lewis structure for each of the following:b. Web practice drawing lewis structures with answers and explanation. Draw the lewis structure, then id shape and polarity. (there are molecules, like o 2, which have unpaired electrons even though they could all be paired, but you can't predict that with lewis structures, so assume they are all paired.)Quick & Easy 5 Steps to Drawing Lewis Structures with Examples
How to draw Lewis Structures a step by step tutorial Middle School
Practice Drawing Lewis Structures Worksheets Worksheets For Kindergarten
3 Ways to Draw Lewis Dot Structures wikiHow
How To Draw Lewis Structures YouTube
How To Draw Lewis Structures A Step By Step Tutorial
How To Draw Lewis Structures A Step By Step Tutorial
Lewis Diagrams Made Easy How to Draw Lewis Dot Structures YouTube
Drawing Lewis Structures Worksheet
Draw Lewis Structure Practice
The Following Procedure Will Give You The Correct Lewis Structure For Any Molecule Or Polyatomic Ion That Has One Central Atom.
You Can Usually Return To This Assignment Anytime During The Course, Even After Its Due Date.
Web The Electrons In The Outermost Shell Are Called The _____ Electrons.
Web The Skeletal Structure Of Ethanethiol Is Shown Below.
Related Post: