Draw The Lewis Structure For H2O
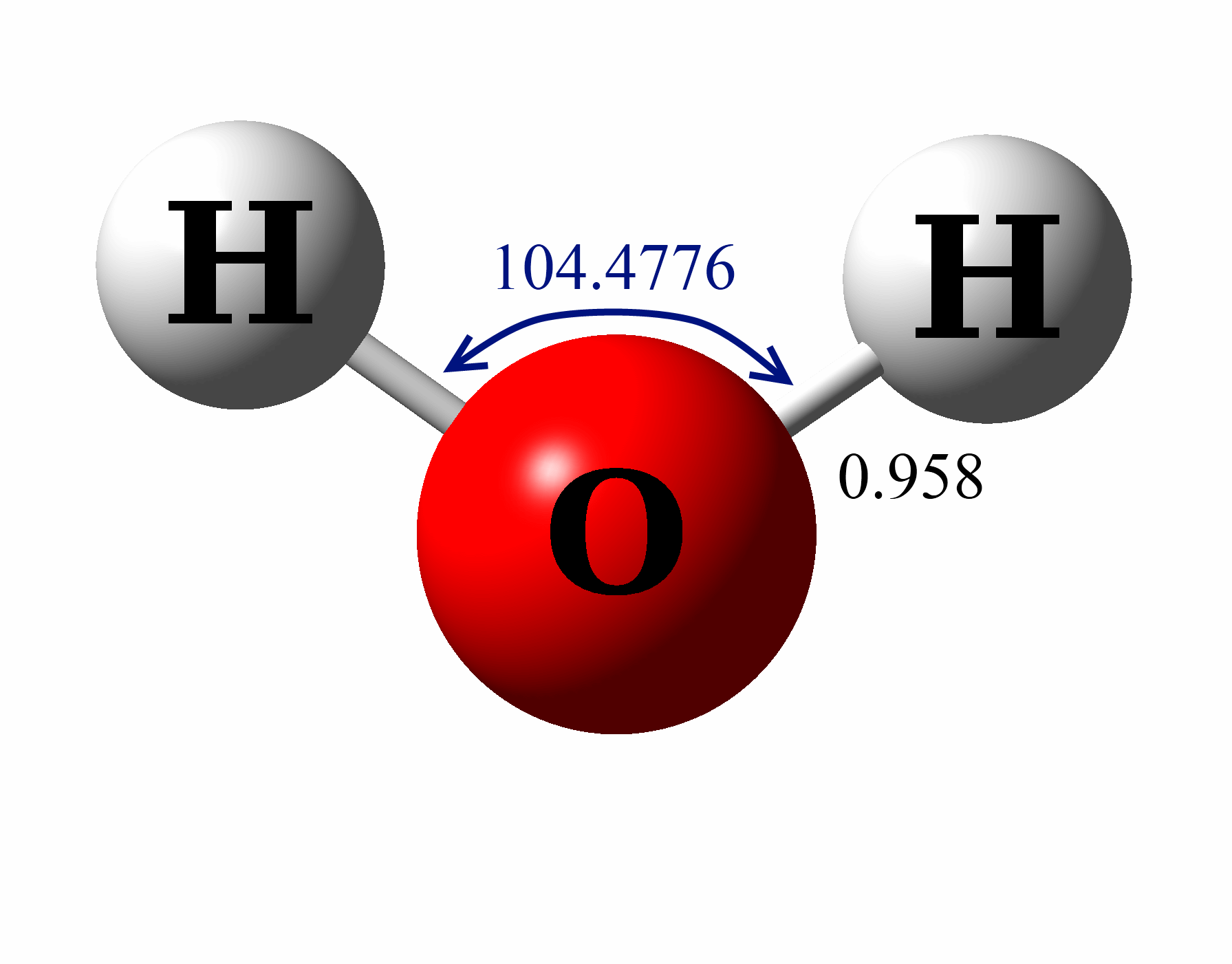
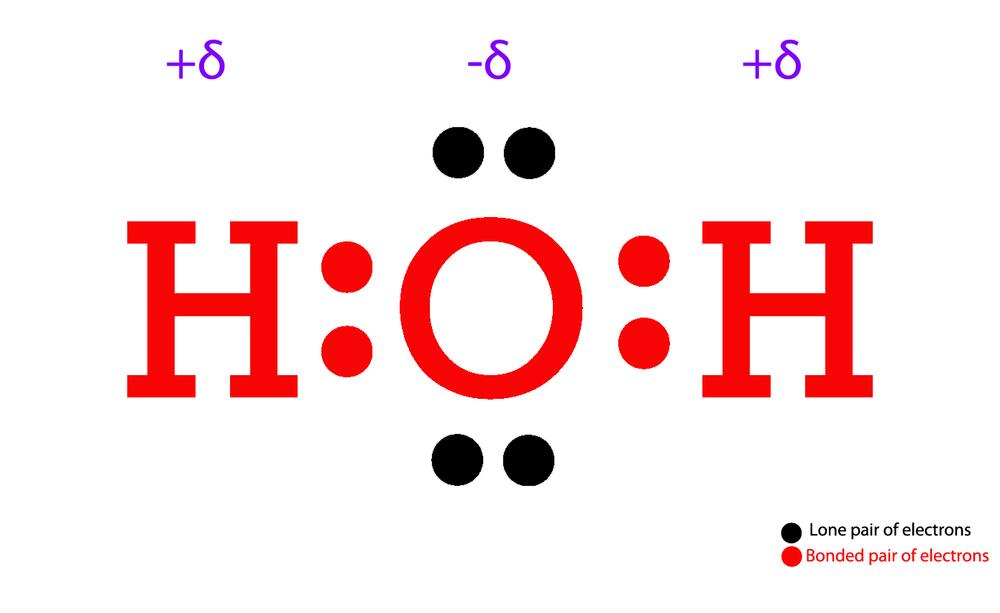
Draw The Lewis Structure For H2O - Thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. Web we can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using lewis dot symbols: See also nh2f lewis structure& characteristics: Web the lewis structure of hydrogen and 2 oxygen atoms shows a total of eight valence electrons participate in the bond formation to form a single triatomic h2o molecule. Place the remaining electrons on the atoms; O has 6 valence electrons, and each h has one. Web what is the lewis structure of water h2o? The structure on the right is the lewis electron structure, or lewis structure, for h2o. Valence electrons are the electrons in the outermost shell of an atom. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. For the h2o structure use the periodic table to find the total number of valence electrons for.more. Molecular geometry of h 2 o. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. We have previously discussed the lewis structures of co2, o3, so2, so3, and more. How to draw lewis structure for h 2 o. Drawing the h2o lewis structure is a simple process that requires a few basic steps: Write lewis symbols for neutral atoms and ions. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Web the structure on the right is the lewis electron structure, or lewis structure, for h 2 o. Web the h2o lewis structure shows the two hydrogen atoms bonded to the oxygen atom, and the two unshared electron pairs on the oxygen atom. For the h2o structure use the periodic table to find the total number of valence electrons for the h2o molecule. Web follow these steps to draw the lewis structure of h2o: Web what is the lewis structure of water h2o? The structure on the right is the lewis electron structure, or lewis structure, for h2o. Molecular geometry of h 2. Connect the atoms with single bonds; The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web the lewis structure of hydrogen and 2 oxygen atoms shows a total of eight valence electrons participate in the bond formation to form a single triatomic h2o molecule. Today we are going. Web i quickly take you through how to draw the lewis structure of water, h2o. Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has a full valence shell of two electrons. Web lewis structure of h2o (or water) contains single bonds between the oxygen (o) atom and each hydrogen (h) atom. Web we can illustrate the. Place the remaining electrons on the atoms; I also go over hybridization, shape and bond angle. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web i quickly take you through how to draw the lewis structure of water, h2o. Count the total valence electrons. How to draw lewis structure for h 2 o. I also go over hybridization, shape and bond angle. Web drawing lewis structures for molecules with one central atom: Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has a full valence shell of two electrons. Web follow these steps to draw the lewis structure of h2o: Hybridization of h 2 o. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. For the h2o structure use the periodic table to find the total number of valence electrons for.more. Place the remaining electrons on the. We have previously discussed the lewis structures of co2, o3, so2, so3, and more. You must arrange 8 electrons in pairs so that o has 8 and each h has two electrons in its valence shell. Web lewis structure of water molecule contains two single bonds around oxygen atom. Web the h2o lewis structure shows the two hydrogen atoms bonded. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. O has 6 valence electrons, and each h has one. The oxygen atom (o) is at the center and it is surrounded by 2 hydrogen atoms (h). Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has a full valence shell. Hybridization of h 2 o. Here, we need to understand how the lewis structure is drawn for the h2o molecule: By the end of this section, you will be able to: Web we can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using lewis dot symbols: Web lewis structure of h2o (or water). I also go over hybridization, shape and bond angle. Web what is the lewis structure of water h2o? Web the lewis structure of hydrogen and 2 oxygen atoms shows a total of eight valence electrons participate in the bond formation to form a single triatomic h2o molecule. Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has. The structure on the right is the lewis electron structure, or lewis structure, for h2o. Web the h2o lewis structure shows the two hydrogen atoms bonded to the oxygen atom, and the two unshared electron pairs on the oxygen atom. Web a video explanation of how to draw the lewis dot structure for water, along with information about the compound including formal charges, polarity, hybrid orbitals, shape, and bond angles. Web i quickly take you through how to draw the lewis structure of water, h2o. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. You must arrange 8 electrons in pairs so that o has 8 and each h has two electrons in its valence shell. With the lewis structure for water (h 2 o) remember that water only needs two valence electrons to have a full outer shell. H2o molecular geometry, lewis structure, shape and bond angles. I also go over hybridization, shape and bond angle. How to draw lewis structure for h 2 o. For h₂o, o must be the central atom. Be sure that you don't use more than the eight valence electrons available. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Web we can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using lewis dot symbols: Web what is the lewis structure of water h2o? How to draw lewis structure for h 2 o.H2O Lewis Structure, Molecular Geometry, and Hybridization
Draw The Lewis Structure Of H2o vrogue.co
Draw Step By Step The Lewis Structure For Water (H2O)
H2O Lewis Structure, Molecular Geometry, and Hybridization
Lewis Structure of Water H2O YouTube
Draw The Lewis Structure Of H2O
Draw Step By Step The Lewis Structure For Water (H2O)
Draw Step By Step The Lewis Structure For Water (H2O) Goimages Solo
12+ Electron Dot Structure Of H2O Robhosking Diagram
Gambar Struktur Lewis H2O pulp
We Have Previously Discussed The Lewis Structures Of Co2, O3, So2, So3, And More.
Let’s Draw And Understand This Lewis Dot Structure Step By Step.
Each Step Of Drawing Lewis Structure Of H 2 O Are Explained In This Tutorial.
Today We Are Going To Learn About The Lewis Structure Of The H2O Molecule Along With Its Molecular Geometry And Shape.
Related Post: